Modelling corneal epithelial wound closure
Corneal wound healing has been the subject of much experimental research. One of the major controversies in this field is the source of epidermal growth factor (EGF) which stimulates the wound healing response. It is known that EGF is present in tears but it is not clear that this is sufficient to account for the observed wound healing response. We analyse a reaction-diffusion model for cell density and EGF concentration in which EGF affects cell movement and division, while cells in turn degrade the EGF. This leads to a novel four-dimensional travelling wave problem which we solve to show that, within the context of this model and the experimental parameters available, the tear source of EGF is insufficient to account for the experimentally observed speed of wound closure. We postulate other possible sources of EGF. We derive an expression for the speed of wound closure which allows us to predict the effects of topical application of EGF. This work appears in publication 44, which won the Sixth Bellman Prize awarded by Mathematical Biosciences.
We extend this model to the more realistic case of multiple cell types (publication 100) with age-structure. We show that this new model is more realistic than the previous model and that it can capture experimentally measured spatio-temporal data on mitotic rates measured during corneal epithelial wound healing. By considering a number of different modeling hypotheses and comparing the predicted results on mitotic rate with the experimental data, we postulate the existence of juxtracrine signalling as a controlling mechanism for cell mitosis and differentiation.
In publication 108, we investigate the effects of electric fields on corneal wound closure by using a moving boundary formalism. The resulting model predicts a linear relation between the wound healing speed and the electric field strength which is robust to variations in critical parameters that are difficult to estimate.
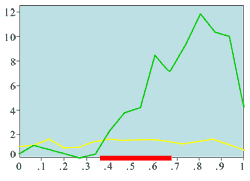
(a) Experimentally measured mitotic rate (ring wound)
|
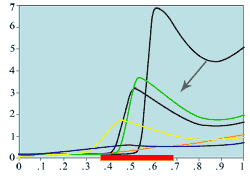
(d) Mitotic rates -
ring/annulus wound
|
|
|
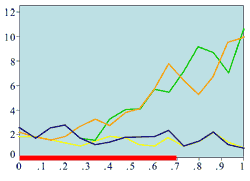
(b) Experimentally measured mitotic rate (large wound)
|
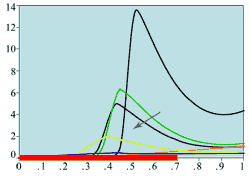
(e) Mitotic rates -
large central wound
|
|
|
(c) Experimentally measured mitotic rate (medium wound)
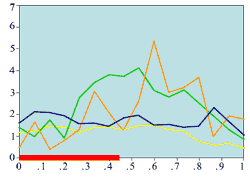
|
(f) Mitotic rates -
medium central wound
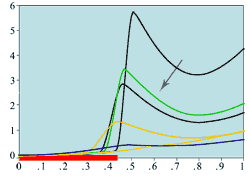
|
Experimental plots of mitotic rates (vertical axes) at various times following wounding of the rat corneal epithelium together with predictions from the model. The horizontal axes are scaled in terms of a typical rat corneal radius, which is approximately 0.4 cm and marked with a red line to denote the initial wound size (0 denotes the central cornea and 1 denotes the outer limbus). In (a) the plots are a control (———) and the mitotic rate at 28 hours after wounding (———); (b) and (c) show the mitotic rates at 24 (———), 48 (———), and 96 (———) hrs after wounding, plus control mitotic rates (———). The plots (d)-(f) are the corresponding numerical simulations of the model. The black lines indicate mitotic rates at 12 and 28 hrs, the colour coding of the other lines are as in the experimental data.
Replotted from Sandvig and Haaskjold (1993), Acta Opthalmol, 71, 39, and Sanvig et al (1994), Acta Opthalmol, 72, 43 [Reproduced from publication 100 with permission]
** Work carried out in collaboration with P.D. Dale, J.A. Sherratt, E.A. Gaffney, S. Tuft, C.D. McCaig, M. Zhao and J.V. Forrester





